 
For any pure substance, the temperature at which melting occurs known as the melting point is a characteristic of that substance. The opposite process, a liquid becoming a solid, is called solidification. The change from solid to liquid usually does not significantly change the volume of a substance. The process of a solid becoming a liquid is called melting (an older term that you may see sometimes is fusion ). Like liquids, gases have no definite shape, but unlike solids and liquids, gases have no definite volume either. A solid has definite volume and shape, a liquid has a definite volume but no definite shape, and a gas has neither a definite volume nor shape. This describes the gas state, which we will consider in more detail elsewhere. If the particles of a substance have enough energy to completely overcome intermolecular interactions, then the particles can separate from each other and move about randomly in space. 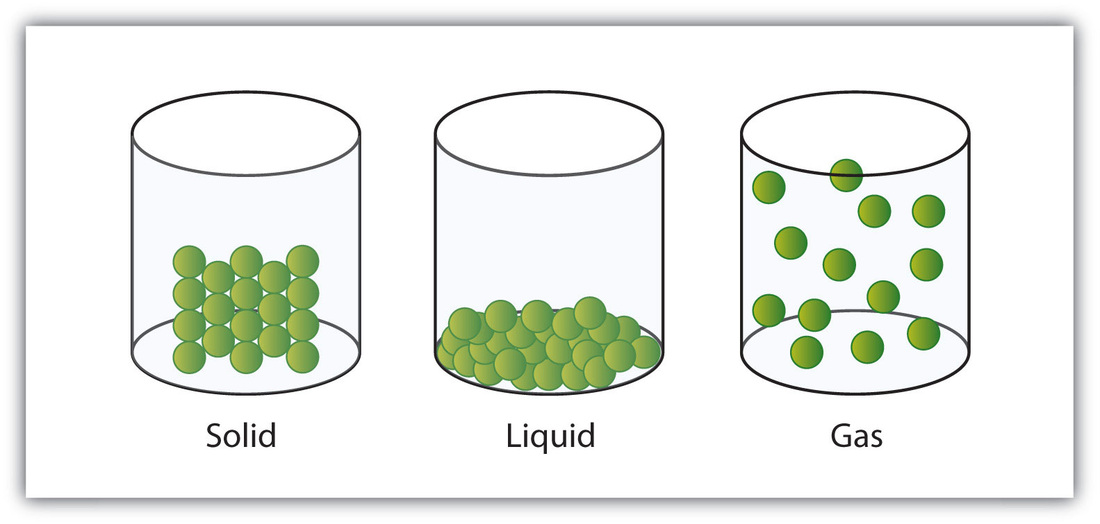
Here you see mercury in its common liquid form. Mercury has a relatively high surface tension, which makes it very unique. Mercury also has an ability to stick to itself (surface tension)-a property that all liquids exhibit. It is the only metal we know of that is liquid at room temperature.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
