 The rise in temperature caused by a given quantity of heat will be different for different substances and depends on the heat capacity of the substance. As energy, in any form is supplied to a system, its molecules begin to rotate, vibrate and translate, which is observable as a rise in temperature. In classical terms, systems at absolute zero have no energy and the atoms or molecules would be close packed together. The first law deals with the conservation of energy, the second law is concerned with the direction in which spontaneous reactions go, and the third law establishes that at absolute zero, all pure substances have the same entropy, which is taken, by convention, to be zero. Alongside this it is important to bear in mind the three laws of thermodynamics. We prefer to consider that the entropy of a system corresponds to the molecular distribution of its molecular energy among the available energy levels and that systems tends to adopt the broadest possible distribution. Secondly, the equation ( ii) defining entropy change does not recognise that the system has to be at equilibrium for it to be valid. First the units of entropy are Joules per Kelvin but the degree of disorder has no units. This more modern approach has two disadvantages. 
However, it is more common today to find entropy explained in terms of the degree of disorder in the system and to define the entropy change, Δ S, as: Where Q is the quantity of heat and T the thermodynamic temperature. Many earlier textbooks took the approach of defining a change in entropy, Δ S, via the equation: Generations of students struggled with Carnot's cycle and various types of expansion of ideal and real gases, and never really understood why they were doing so. The concept of entropy emerged from the mid-19th century discussion of the efficiency of heat engines. Entropy is a thermodynamic property, like temperature, pressure and volume but, unlike them, it can not easily be visualised. Thermodynamic properties depend on the current state of the system but not on its previous history and are either extensive - their values depend on the amount of substance comprising the system, eg volume - or intensive - their values are independent of the amount of substance making up the system, eg temperature and pressure. Thermodynamics deals with the relation between that small part of the universe in which we are interested - the system - and the rest of the universe - the surroundings. \(\Delta S_\).Entropy is dynamic - something which static scenes don't reflect If the happening process is at a constant temperature then entropy will be Furthermore, it includes the entropy of the system and the entropy of the surroundings.īesides, there are many equations to calculate entropy:ġ. Also, scientists have concluded that in a spontaneous process the entropy of process must increase. Moreover, the entropy of solid (particle are closely packed) is more in comparison to the gas (particles are free to move). Entropy FormulaĮntropy is a thermodynamic function that we use to measure uncertainty or disorder of a system. In addition, some microscope process is reversible. 
Besides, some other example of changeable phase is the melting of metals. On the other hand, blowing a building, frying an egg is an unalterable change. 
Moreover, when the process is unalterable then the entropy will increase.įor example, watching a movie is a changeable process because you can watch the movie from backward. Also, even when the cyclic process is changeable then the entropy will not change. The second law of thermodynamics says that every process involves a cycle and the entropy of the system will either stay the same or increase. Get the huge list of Physics Formulas here The Second Law of Thermodynamics Furthermore, the more you increase the ball the more ways it can be arranged. So, now you can arrange the balls in two ways. 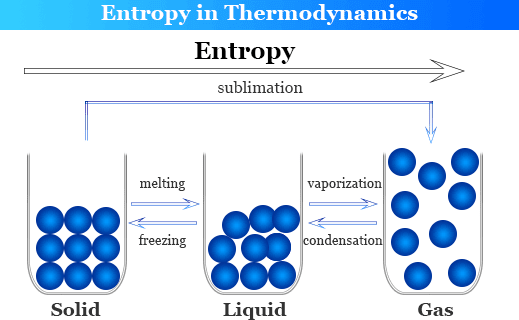
After some time you put another ball on the table. Moreover, the question here is in how many ways you can arrange this ball? The answer is one. In another example, you grab a ball and put it on a table. So, what will happen next? We all know that the smell will spread in the entire room and the perfume molecule will eventually fill the room. Suppose you sprayed perfume in one corner of the room. Furthermore, we can understand it more easily with the help of an example. Moreover, the higher the entropy the more disordered the system will become. Entropy refers to the number of ways in which a system can be arranged. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
